 |
||
| 7625 West New York Street | ||
| Indianapolis, Indiana 46214-4911 USA | ||
| Telephone: (317) 273-6960 | ||
| Facsimile: (317) 273-6979 | ||
| Order Desk Telephone: (800) 401-1723 | ||
| Order Desk Facsimile: (800) 515-9254 | ||
| E-mail: info@floteco2.com | ||
 |
||
| 7625 West New York Street | ||
| Indianapolis, Indiana 46214-4911 USA | ||
| Telephone: (317) 273-6960 | ||
| Facsimile: (317) 273-6979 | ||
| Order Desk Telephone: (800) 401-1723 | ||
| Order Desk Facsimile: (800) 515-9254 | ||
| E-mail: info@floteco2.com | ||
WHY HIGH STRENGTH, ANODIZED, ALUMINUM ALLOY IS SUITABLE FOR USE IN OXYGEN REGULATORS
CONFIDENTIAL
On rare occasions, a fire will occur in an oxygen system. The incidence of oxygen system fires is low, often with the probability in the one-in-a-million range, even when rather incompatible systems are involved. Fires have been reported in stainless steel ball valves used in oxygen transfilling stations, space capsules, in home liquid oxygen stainless steel tank accessories, flange seals in large steel pipe oxygen systems, and even the waxed mustaches of airplane pilots breathing pure oxygen.
Oxygen’s ability to chemically combine with other materials is the source of its value. It is also the source of its hazard. Ideally; cylinders, piping, valves, regulators, and flow meters would be constructed entirely of non-flammable materials, such as gold, silver, and oxide ceramics (aluminum oxide). Practical, non-leaking systems, however, require resilient sealing materials made of plastics or synthetic rubbers, which, unfortunately, are less resistant to ignition than metals. In addition, the presence of contaminants such as metallic or plastic particles, or hydrocarbons can cause ignition under certain conditions.
All fires require three elements, sometimes called the fire triangle.
Even when all 3 elements are present, fires are rare in oxygen systems, because these 3 elements, of and by themselves, are not sufficient to cause the materials to burn.
QUESTION: What are the sources of ignition in oxygen systems?
ANSWER:
QUESTION: Why are you telling us this?
ANSWER: In the last year, we at Flotec, have been asked by a few concerned users of aluminum oxygen regulators to respond to the question:
"Are Aluminum Oxygen Regulators Safe?"
Flotec’s answer is "Some are." Some are not."
QUESTION: Why did they ask?
ANSWER: In the last 20 years 24 instances of ignition in oxygen regulators have been reported as follows:
LSP/Allied Healthcare 18
Puritan Bennett 2
Sunrise 1
Western 1
Veriflow 1
Unknown 1
_____
TOTAL 24
The FDA and NIOSH published an advisory in February 1999 stating "that the aluminum in these regulators was a major factor in both the ignition and severity of the fire although there are likely other contributing factors".
This statement is not supported by the Bell Evaluation Laboratory report, which examined two of the LSP/Allied Healthcare regulators, which ignited at the Houston Texas fire department. Their report stated that:
"The ignition of non-metallic contaminate within the regulator by adiabatic compression or metallic particle impingement during initial pressurization and a subsequent kindling chain event occurred."
-- OR --
"Metallic contaminate embedded in the Teflon seal impacted the seat ring and ignited the Teflon seal during initial pressurization/seat closure and a subsequent kindling chain event occurred."
"Both of these conditions are a result of particulate contaminate entering the regulator through a screen which is inadequate to keep materials generated by the associated equipment (oxygen cylinder valve stem) from entering the regulator. The aluminum regulator construction makes the result of such ignition events catastrophic due to the flammability of the aluminum itself. A better design would include sintered bronze inlet filters (not a screen) and brass regulator bodies."
Bell’s report shows clearly that ignition did not begin in the anodized aluminum body but in the contaminants. The ignition would not have been prevented nor delayed by the use of material other than aluminum, such as brass. Undoubtedly the severity of the fire would have been reduced by brass; however, it is likely that molten brass slag would have been ejected through the DISS check valves as the valve poppet seals burned away.
It is clear that eliminating one of the three elements of the fire triangle can prevent the regulator fires. Merely substituting brass for aluminum will remove only one element. Although brass will not burn, it will melt.
QUESTION: Is it possible to design and build a regulator that is absolutely fireproof?
ANSWER: No.
To obtain zero leakage in a high-pressure device, elastomeric seals must be used. Seal materials are plastic (Kel F, Teflon, Nylon, etc.) or synthetic rubber which will ignite and burn much more readily than metals. The risk can be reduced by proper design, selection of materials, and correct maintenance and operation by the user. If the seals of a pressure device burn, the supporting metal will melt. If the seal is destroyed and hot, high-pressure gas escapes, hot metal slag may be ejected. This molten slag is the source of most injuries or damage.
QUESTION: Is it possible to design and build an aluminum oxygen regulator which, when maintained and used correctly, reduces the risk of fire to almost zero?
ANSWER: Yes
Flotec, Inc. has done so.
Section 7 of ASTM Guide G88-90 lists the principles for improving fire resistance. The Flotec design, which is currently being copied by others, has never been involved in an incident in which its regulators incurred ignition. This includes its older designs made from 1982 to 1994, when the new, all metal, unibody window designs were introduced.
Since all oxygen systems contain two of the elements required to cause a fire (fuel and oxidant) the priority should be to eliminate the third - ignition.
Only two ignition sources are significant.
1. Heat caused by adiabatic (isotropic) compression
2. High speed impacts of a particle larger than 100 micron
The solution is obvious - Eliminate ignition by:
- Filter out particles larger than 100 microns - Flotec uses a depth type sintered bronze filter, which traps particles larger than 20 microns. The filter is pressed into the ID of the brass retainer and then installed into the regulator body. This prevents the filter from being damaged or restricting flow when squeezed into position by designs that pinch the filter between the aluminum body of the regulator and the retainer which is torqued into position during assembly.
- Prevent adiabatic compression from raising the temperature of the oxygen to the ignition point of the seals or metal.
Flotec regulators use a brass relief valve/pressure-reducing cartridge, which prevents the internal pressure from exceeding 95 psi. The modulating piston moves forward and shuts off the inlet orifice if the pressure exceeds 57 psi when inlet pressure is 3000 psi. The relief valve piston reacts instantly and dumps the incoming gas should pressure exceed 90 to 95 psi.
Please see drawing attached which describes this device.
Flotec regulator structural parts are made heat-treated aluminum alloys, which greatly enhances the resistance to ignition in a pure oxygen environment.
All aluminum parts are "anodized", a process which electrochemically converts the surface of the aluminum to aluminum oxide - a material which cannot burn since it is already completely oxidized.
The high-pressure oxygen inlet conduit in Flotec regulators is made of brass, which is superior to aluminum in its resistance to burning.
Small, heat-resisting seals are used throughout. Seals are recessed in the metal components to limit exposure to the gas.
Internal cavities are kept small. No thin sections are allowed. All fins and burrs are removed.
QUESTION: Does it work?
ANSWER: It has on the 449,000 Flotec regulators in service and we’re still counting.
QUESTION: Have attempts been made to deliberately cause fires in aluminum oxygen regulators?
ANSWER: YES. In 1987, aluminum oxygen regulators were submitted to NASA’s White Sands
Proving Grounds. Their assignment was to cause ignition within the regulators. The design of the relief valve/pressure-reducing cartridge was almost identical to Flotec’s latest design shown on Exhibit # B.
After 5 days, technicians were still unable to cause ignition. As a last resort grease and salad oil were introduced in the regulators. Still, no ignition occurred. The relief valve/pressure-reducing cartridge successfully limits the internal gas pressure to about 100 psi, even when the cylinder valve is opened quickly. Heat arising from adiabatic compression is limited to such low levels that ignition is virtually impossible. With the exception of LSP, Allied Health Care and Contemporary Products, Inc. (CPI), which has no relief valve at all) most of the aluminum unibody regulators being produced contain a dual-piston pressure reducing/relief valve cartridge. See Exhibit # B for details on its operation. The modulating piston reduces the high pressure of the gas entering the regulator from 2000 psi to a nominal 50 psi. Should this not occur, the relief valve piston (which encircles the inlet orifice), opens instantly if the pressure approaches 100 psi and vents the gas to atmosphere. Adiabatic compression of oxygen to 100 psi, produces so little heat that it can almost be ignored as a possible source of ignition.
This leaves only particle impact as a possible source of ignition. Literature indicates that only particles in the 100 to 800 micron range impacting at very high velocities in pressurized oxygen have enough kinetic energy to cause ignition. Preventing particles larger than 20 microns from entering the regulator, will almost entirely eliminate the risk of particle impact ignition in the regulator, as most aluminum regulators are anodized inside and out. Anodizing electro chemically converts the surface of aluminum to aluminum oxide, an extremely hard material that is incapable of burning, since it is already completely oxidized.
Exhibit A
Safe Practices for Handling and Operating Oxygen Equipment
Oxygen used in the medical profession can be very hazardous. Although oxygen does not burn, it does support combustion. A material which will not burn in air may burn in high pressure pure oxygen - such as the metal in oxygen regulators or cylinders. Comprehensive guidelines and training on safe practices for handling oxygen are available form several sources listed at the end of this section. Some general guidelines for minimizing the chance of fire are provided below:
Storage, Maintenance and Handling:
Use:
Additional information, guidance and training regarding oxygen and fire safety can be obtained from a number of sources, including the following organizations:
Exhibit B CONFIDENTIAL
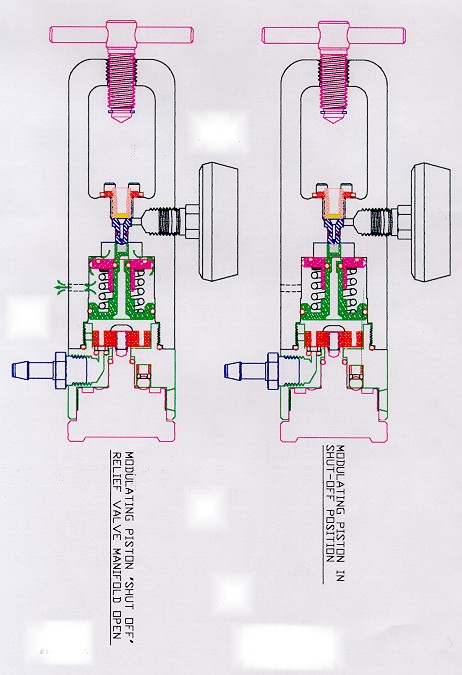
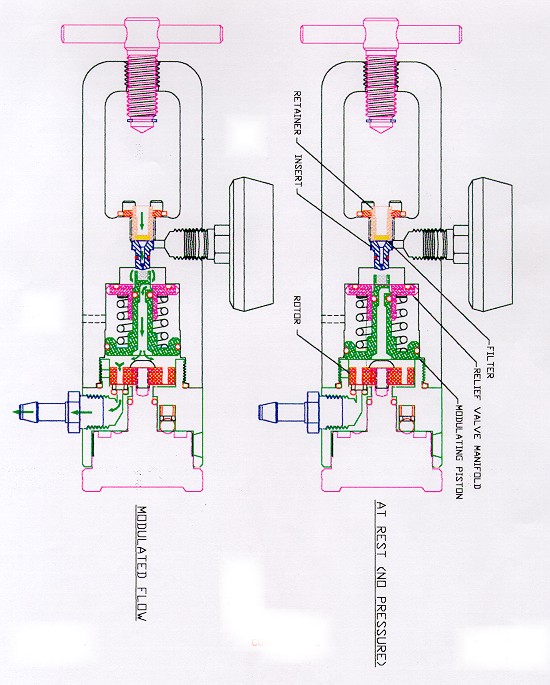
Exhibit C
The facts in this bulletin are taken from:
TB-AluminumRegulator.doc(06-09-99)